Transparent MedTech logistics
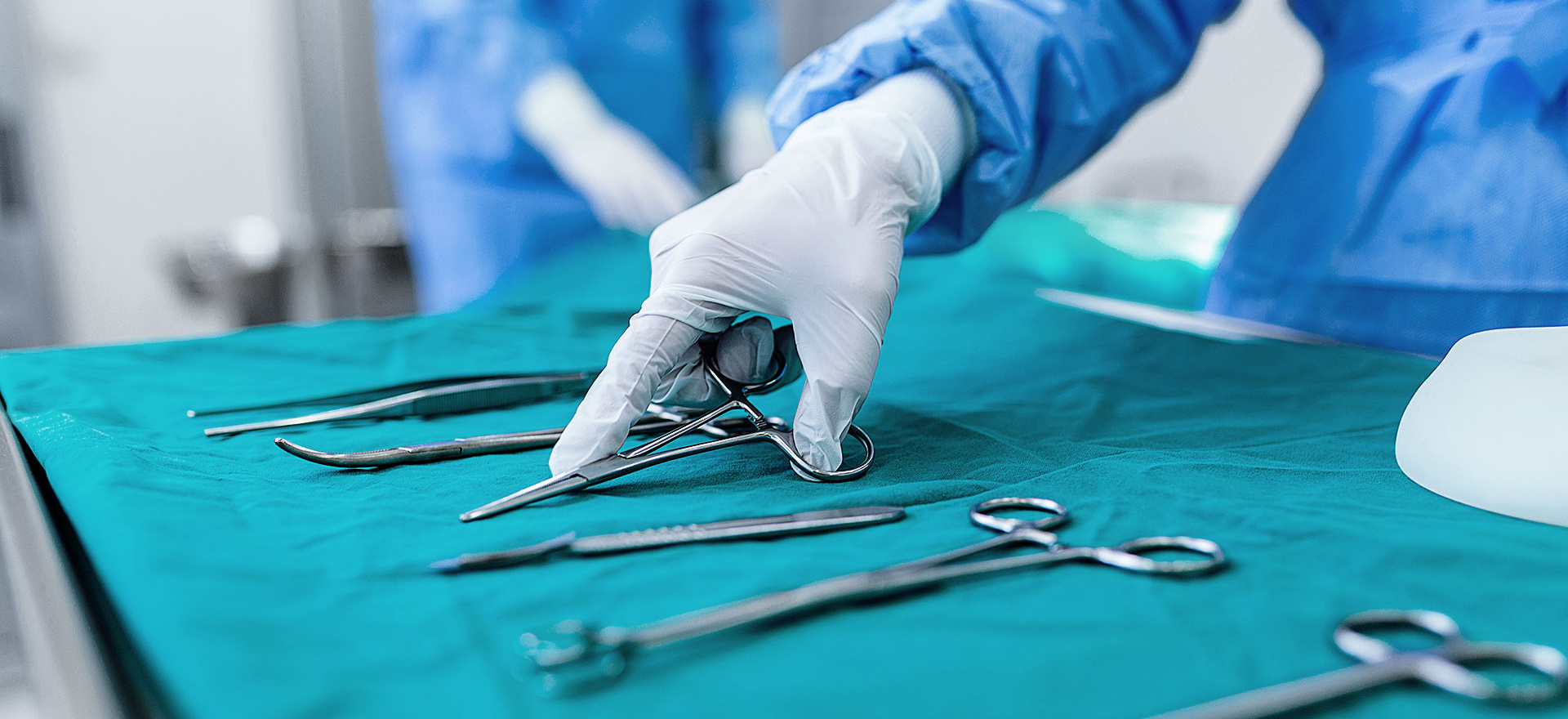
Always state of the art.
It takes certain knowledge and training to handle medical instruments correctly. The Medical Device Regulation (MDR) and the EU’s In Vitro Diagnostic Regulation (IVDR) set out high regulatory standards. We optimise your supply chain and prepare you for the industry’s global production flows while offering maximum transparency – enabling you to always stay on top of things.
- International transportation: We deliver medical products and diagnostics quickly and reliably to your intended destination. By land. By sea. By air.
- Consolidating resources: Our adaptable capacities and trained personnel are here to operate your central distribution centre for you.
- Returns and reprocessing: Our full-service offer encompasses the sterile processing of medical equipment, including routine quality checks.
Our logistics services perfectly complement the following product groups:
Coffee
Food
Food-
supplments
Wine
Our solutions
Certified logistics concepts as a customised or standardised industry option – this is what we offer the MedTech sector. ISO 13485-certified.
Why FIEGE?
More than 25 years of experience
We have been active in the healthcare sector for over two decades and are familiar with the industry’s challenges.
Certified quality management
We ensure quality. With industry-specific certifications and regular audits.
Fast and reliable
As a family-run business, we ensure swift decision-making and process consistency.
International network
Our Europe-wide network is tightly connected, enabling international coverage.
Many healthcare businesses have relied on our expertise for years.
















